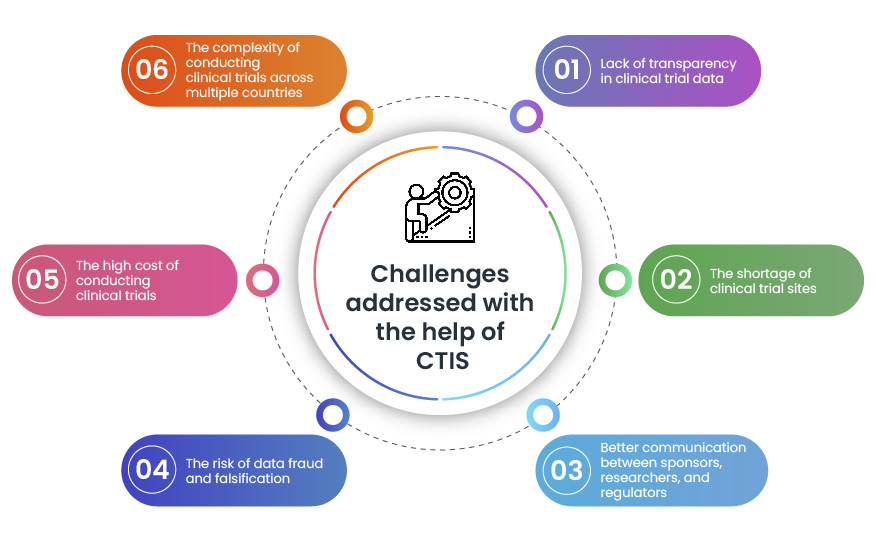
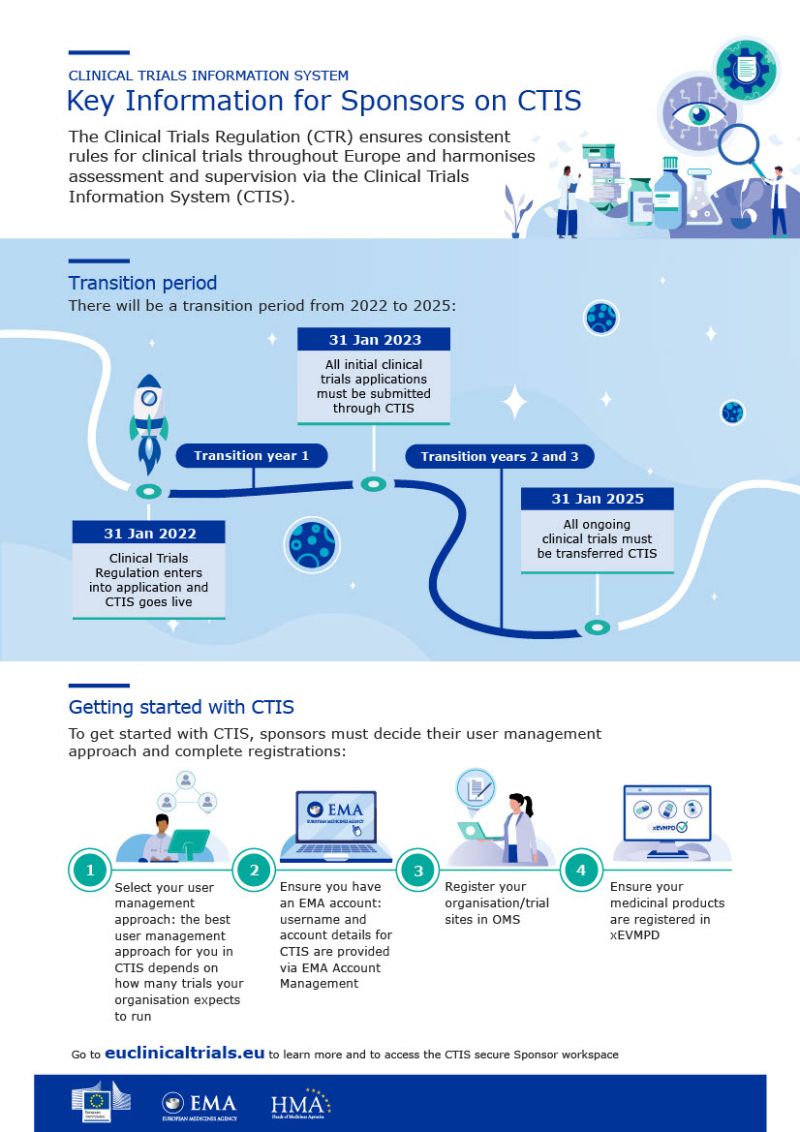
.jpg)
The Clinical Trial Information System (CTIS) and Data Privacy: A Guide for Clinical Trial Sponsors | Iliomad

Managing the New EU Clinical Trials Regulation 536/2014 – Guidance for Navigating the Clinical Trial Information System (CTIS)

ANSM Agence nationale de sécurité du médicament et des produits de santé sur LinkedIn : Use of Clinical Trials Information System becomes mandatory for new…

Webinaire Clinical Trials Information System (CTIS): Préparation à l'utilisation obligatoire de la Régulation des Essais Cliniques à compter du 31/01/2023